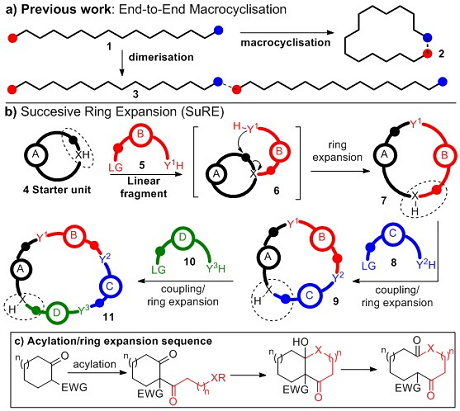
Important applications across the physical sciences rely on the synthesis of functionalised macrocycles. At present, macrocycles are typically made via the end-to-end cyclisation of a linear precursor, a notoriously difficult and unpredictable transformation; in particular, achieving macrocyclisation (1 → 2, Figure 1a) rather than dimerisation (1 → 3) is a major challenge. The most common strategy used to combat this is to perform the reactions at high-dilution but while successful in many cases, such protocols are generally highly substrate dependent and impractical for large scale synthesis.
Successive Ring Expansion (SuRE for short) is designed to improve the efficiency of macrocycle synthesis by completely avoiding end-to-end macrocyclisation. It is based on the sequential insertion of linear fragments into existing cyclic systems, by coupling a cyclic compound (4) to a linear fragment 5, which can thenrearrange (6), initiating ring expansion, forming a ring enlarged product 7. A key design feature is the replication of the functionality in the cyclic starter unit in the ring-expanded product (circled), as this means that the same coupling/ring expansion sequence can be repeated, allowing further iterations to be performed in the same way (7 → 9; 9 → 11). SuRE can incorporate a range of linear fragments and can theoretically be repeated indefinitely, meaning macrocycles of virtually any ring size and composition are potentially accessible.
Successive Ring Expansion (SuRE for short) is designed to improve the efficiency of macrocycle synthesis by completely avoiding end-to-end macrocyclisation. It is based on the sequential insertion of linear fragments into existing cyclic systems, by coupling a cyclic compound (4) to a linear fragment 5, which can thenrearrange (6), initiating ring expansion, forming a ring enlarged product 7. A key design feature is the replication of the functionality in the cyclic starter unit in the ring-expanded product (circled), as this means that the same coupling/ring expansion sequence can be repeated, allowing further iterations to be performed in the same way (7 → 9; 9 → 11). SuRE can incorporate a range of linear fragments and can theoretically be repeated indefinitely, meaning macrocycles of virtually any ring size and composition are potentially accessible.
More deatils on this concept can be found in our first publication arising from this project, see: Angew. Chem. Int. Ed. 2015, 54, 15794 –15798. In this paper, the SuRE concept is validated using a telescoped two-step sequence, to generate macrocyclic lactams and lactones from cyclic β-keto esters (Figure 1c).
See also: Ring expansion approach to medium-sized lactams and analysis of their medicinal lead-like properties
L. G. Baud, M. A. Manning, H. L. Arkless, T. C. Stephens, W. P. Unsworth, Chem. Eur. J. 2017, 23, 2225.
Synthesis of Cyclic Peptide Mimetics by the Successive Ring Expansion of Lactams
T. C. Stephens, M. Lodi, A. Steer, Y. Lin, M. Gill, W. P. Unsworth, Chem. Eur. J. 2017 doi: 10.1002/chem.201703316
See also: Ring expansion approach to medium-sized lactams and analysis of their medicinal lead-like properties
L. G. Baud, M. A. Manning, H. L. Arkless, T. C. Stephens, W. P. Unsworth, Chem. Eur. J. 2017, 23, 2225.
Synthesis of Cyclic Peptide Mimetics by the Successive Ring Expansion of Lactams
T. C. Stephens, M. Lodi, A. Steer, Y. Lin, M. Gill, W. P. Unsworth, Chem. Eur. J. 2017 doi: 10.1002/chem.201703316
