2024
83.
Expanding the scope of the Successive Ring Expansion strategy for macrocycle and medium-sized ring synthesis: unreactive and reactive lactams
Z. Yang, M. Arnoux, D. Hazelard, O. Hughes, J. Nabarro, A. C. Whitwood, M. A Fascione, C. D. Spicer, P. Compain, W. P. Unsworth. Org. Biomol. Chem., 2024,
https://doi.org/10.1039/D4OB00285G
Expanding the scope of the Successive Ring Expansion strategy for macrocycle and medium-sized ring synthesis: unreactive and reactive lactams
Z. Yang, M. Arnoux, D. Hazelard, O. Hughes, J. Nabarro, A. C. Whitwood, M. A Fascione, C. D. Spicer, P. Compain, W. P. Unsworth. Org. Biomol. Chem., 2024,
https://doi.org/10.1039/D4OB00285G

82.
The genomic and enzymatic basis for iridoid biosynthesis in cat thyme.
S. J. Smit, S. Ayten, B. A. Radzikowska, J. P. Hamilton, S. Langer, W. P. Unsworth, T. R. Larson, C. R. Buell, B. R. Lichman, Plant J. 2024 (Teucrium marum).
https://doi.org/10.1111/tpj.16698
The genomic and enzymatic basis for iridoid biosynthesis in cat thyme.
S. J. Smit, S. Ayten, B. A. Radzikowska, J. P. Hamilton, S. Langer, W. P. Unsworth, T. R. Larson, C. R. Buell, B. R. Lichman, Plant J. 2024 (Teucrium marum).
https://doi.org/10.1111/tpj.16698
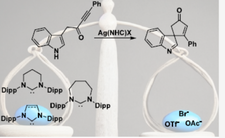
81.
Silver–N-Heterocyclic Carbenes in π–Activation: Synergistic Effects between the Ligand Ring Size and the Anion
A. Frith, A. K. Clarke, A. Heyam, J. M. Lynam, P. D. Newman, W. P. Unsworth, C. E. Willans.
Organometallics 2024, 43, 5, 598–604
Silver–N-Heterocyclic Carbenes in π–Activation: Synergistic Effects between the Ligand Ring Size and the Anion
A. Frith, A. K. Clarke, A. Heyam, J. M. Lynam, P. D. Newman, W. P. Unsworth, C. E. Willans.
Organometallics 2024, 43, 5, 598–604
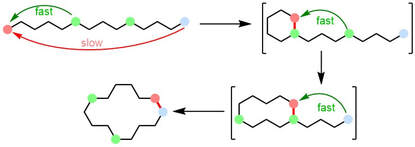
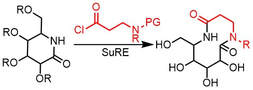
80.
A Modular Strategy for the Synthesis of Macrocycles and Medium-Sized Rings via Cyclization/Ring Expansion Cascade Reactions.
I. Zalessky, J. M. Wootton, J. K. F. Tam, D. E. Spurling, W. C. Glover-Humphreys, J. R. Donald, W. E. Orukotan, L. C. Duff, B. J. Knapper, A. C. Whitwood, T. F. N. Tanner, A. H. Miah, J. M. Lynam and W. P. Unsworth
J. Am. Chem. Soc. 2024, doi.org/10.1021/jacs.4c00659
A Modular Strategy for the Synthesis of Macrocycles and Medium-Sized Rings via Cyclization/Ring Expansion Cascade Reactions.
I. Zalessky, J. M. Wootton, J. K. F. Tam, D. E. Spurling, W. C. Glover-Humphreys, J. R. Donald, W. E. Orukotan, L. C. Duff, B. J. Knapper, A. C. Whitwood, T. F. N. Tanner, A. H. Miah, J. M. Lynam and W. P. Unsworth
J. Am. Chem. Soc. 2024, doi.org/10.1021/jacs.4c00659
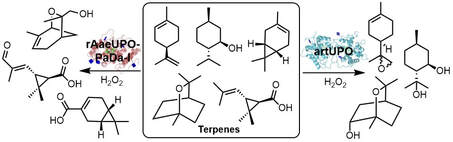
79.
Complementary specificity of unspecific peroxygenases enables access to diverse products from terpene oxygenation.
B. Melling, T. Mielke, A. C. Whitwood, T. J. C. O’Riordan, N. Mulholland, J. Cartwright, W. P. Unsworth, G. Grogan
Chem Catalysis 2024, 4, 100889
Complementary specificity of unspecific peroxygenases enables access to diverse products from terpene oxygenation.
B. Melling, T. Mielke, A. C. Whitwood, T. J. C. O’Riordan, N. Mulholland, J. Cartwright, W. P. Unsworth, G. Grogan
Chem Catalysis 2024, 4, 100889
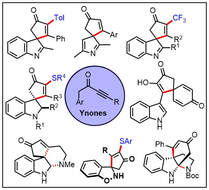
78.
Ynones in dearomative spirocyclisation processes; a review.
R. J. K. Taylor, W. P. Unsworth
Tetrahedron Chem 2024, 9, 100055
Ynones in dearomative spirocyclisation processes; a review.
R. J. K. Taylor, W. P. Unsworth
Tetrahedron Chem 2024, 9, 100055
2023

77.
Divergent Cascade Ring Expansion Reactions of Acryloyl Imides.
W. E. Orukotan, K. Y. Palate, B. Pogrányi, P. Bobinski, R. G. Epton, L. Duff, A. C. Whitwood, G. Grogan, J. M. Lynam, W. P. Unsworth.
Chem. Eur. J. 2023, 29, e202303270
Divergent Cascade Ring Expansion Reactions of Acryloyl Imides.
W. E. Orukotan, K. Y. Palate, B. Pogrányi, P. Bobinski, R. G. Epton, L. Duff, A. C. Whitwood, G. Grogan, J. M. Lynam, W. P. Unsworth.
Chem. Eur. J. 2023, 29, e202303270
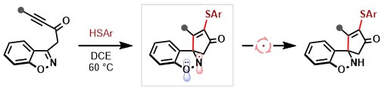
76.
Radical Dearomatising Spirocyclisation of Benzisoxazole-Tethered Ynones.
N. Inprung, A. C. Whitwood, R. J. K. Taylor, M. J. James, W. P. Unsworth
Eur. J. Org. Chem. 2023, e202300603
Radical Dearomatising Spirocyclisation of Benzisoxazole-Tethered Ynones.
N. Inprung, A. C. Whitwood, R. J. K. Taylor, M. J. James, W. P. Unsworth
Eur. J. Org. Chem. 2023, e202300603
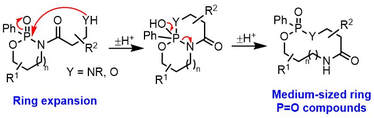
75.
Ring expansion reactions of P=O-containing Molecules.
Z. Yang, J. K. F. Tam, J. M. Wootton, J. M. Lynam, W. P. Unsworth,
Chem. Commun. 2023, 59, 7927–7930
Ring expansion reactions of P=O-containing Molecules.
Z. Yang, J. K. F. Tam, J. M. Wootton, J. M. Lynam, W. P. Unsworth,
Chem. Commun. 2023, 59, 7927–7930
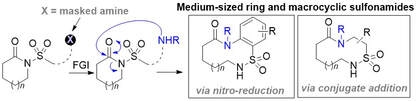
74.
Ring Expansion Strategies for the Synthesis of Medium Sized Ring and Macrocyclic Sulfonamides.
Z. Yang, I. Zalessky, R. G. Epton, A. C. Whitwood, J. M. Lynam, W. P. Unsworth
Angew. Chem., Int. Ed. 2023, 62, e202217178
Ring Expansion Strategies for the Synthesis of Medium Sized Ring and Macrocyclic Sulfonamides.
Z. Yang, I. Zalessky, R. G. Epton, A. C. Whitwood, J. M. Lynam, W. P. Unsworth
Angew. Chem., Int. Ed. 2023, 62, e202217178
73.
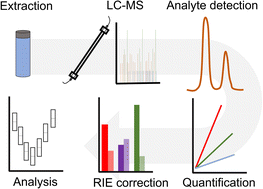
Overcoming the lack of authentic standards for the quantification of biogenic secondary organic aerosol markers.
D. J. Bryant, A. W. Mayhew, K. L. Pereira, S. H. Budisulistiorini, C. Prior, W. P. Unsworth, D. O. Topping, A. R. Rickard J. F. Hamilton
Environ. Sci.: Atmos., 2023, https://doi.org/10.1039/D2EA00074A
Overcoming the lack of authentic standards for the quantification of biogenic secondary organic aerosol markers.
D. J. Bryant, A. W. Mayhew, K. L. Pereira, S. H. Budisulistiorini, C. Prior, W. P. Unsworth, D. O. Topping, A. R. Rickard J. F. Hamilton
Environ. Sci.: Atmos., 2023, https://doi.org/10.1039/D2EA00074A
72.
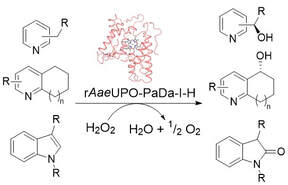
Preparative-Scale Biocatalytic Oxygenation of N-Heterocycles with a Lyophilized Peroxygenase Catalyst.
B. Pogrányi, T. F. Mielke, A. Díaz-Rodriguez, J. Cartwright, W. P Unsworth, G. Grogan
Angew. Chem., Int. Ed. 2023, 62, e202214759
Preparative-Scale Biocatalytic Oxygenation of N-Heterocycles with a Lyophilized Peroxygenase Catalyst.
B. Pogrányi, T. F. Mielke, A. Díaz-Rodriguez, J. Cartwright, W. P Unsworth, G. Grogan
Angew. Chem., Int. Ed. 2023, 62, e202214759
2022
71.
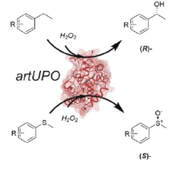
Comparing the catalytic and structural characteristics of a ‘short’ unspecific peroxygenase (UPO) expressed in P. pastoris and E. coli.
W. X. Q. Robinson, T. Mielke, B. Melling, A. Cuetos, A. Parkin, W. P. Unsworth, J. Cartwright, G. Grogan
ChemBioChem 2022, https://doi.org/10.1002/cbic.202200558
Comparing the catalytic and structural characteristics of a ‘short’ unspecific peroxygenase (UPO) expressed in P. pastoris and E. coli.
W. X. Q. Robinson, T. Mielke, B. Melling, A. Cuetos, A. Parkin, W. P. Unsworth, J. Cartwright, G. Grogan
ChemBioChem 2022, https://doi.org/10.1002/cbic.202200558
70.
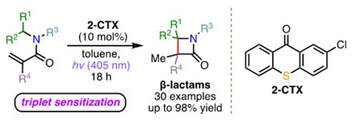
Visible-Light Mediated Energy Transfer Enables the Synthesis of β-Lactams via Intramolecular Hydrogen Atom Transfer.
M. J. Oddy, D. A. Kusza, R. G. Epton, J. M. Lynam, W. P. Unsworth, W. F. Petersen
Angew. Chem., Int. Ed. 2022, 2022, 61, e202213086
Visible-Light Mediated Energy Transfer Enables the Synthesis of β-Lactams via Intramolecular Hydrogen Atom Transfer.
M. J. Oddy, D. A. Kusza, R. G. Epton, J. M. Lynam, W. P. Unsworth, W. F. Petersen
Angew. Chem., Int. Ed. 2022, 2022, 61, e202213086
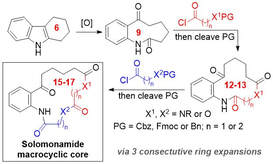
69.
A consecutive ring expansion strategy towards the macrocyclic core of the solomonamide natural products.
Z. Yang, C. R. B. Swanson, W. P. Unsworth
Synlett 2022 DOI: 10.1055/a-1932-9717
A consecutive ring expansion strategy towards the macrocyclic core of the solomonamide natural products.
Z. Yang, C. R. B. Swanson, W. P. Unsworth
Synlett 2022 DOI: 10.1055/a-1932-9717
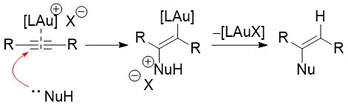
68.
DFT Studies of Au(I) Catalysed Reactions: Anion Effects and Reaction Selectivity.
R. G. Epton, W. P. Unsworth, J. M. Lynam
Isr. J. Chem. 2022 e202200033
DFT Studies of Au(I) Catalysed Reactions: Anion Effects and Reaction Selectivity.
R. G. Epton, W. P. Unsworth, J. M. Lynam
Isr. J. Chem. 2022 e202200033
67.
Biocatalytic activation of diazirines for carbene-transfer reactions
B. Pogrányi, W. P. Unsworth, G. Grogan
Chem., 2022 8, 1801-1804
Biocatalytic activation of diazirines for carbene-transfer reactions
B. Pogrányi, W. P. Unsworth, G. Grogan
Chem., 2022 8, 1801-1804
66.
Synthesis of medium-ring lactams and macrocyclic peptide mimetics via conjugate addition/ring expansion cascade reactions.
K. Y. Palate, Z. Yang, A. C. Whitwood, W. P. Unsworth
RSC Chem. Biol., 2022 ASAP
Synthesis of medium-ring lactams and macrocyclic peptide mimetics via conjugate addition/ring expansion cascade reactions.
K. Y. Palate, Z. Yang, A. C. Whitwood, W. P. Unsworth
RSC Chem. Biol., 2022 ASAP
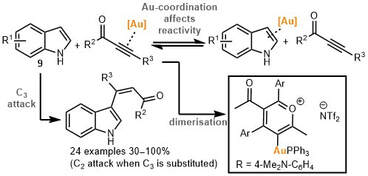
65.
Selectivity, Speciation, and Substrate Control in the Gold-Catalyzed Coupling of Indoles and Alkynes.
R. G. Epton, W. P. Unsworth, J. M. Lynam
Organometallics 2022 ASAP
Selectivity, Speciation, and Substrate Control in the Gold-Catalyzed Coupling of Indoles and Alkynes.
R. G. Epton, W. P. Unsworth, J. M. Lynam
Organometallics 2022 ASAP
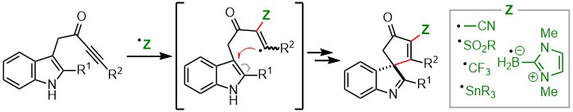
64.
Indole-ynones as Privileged Substrates for Radical Dearomatizing Spirocyclization Cascades.
N. Inprung, H. E. Ho, J. A. Rossi-Ashton, R. G. Epton, A. C. Whitwood, J. M. Lynam, R. J. K. Taylor, M. J. James, W. P. Unsworth
Org. Lett. 2022, 24, 668–674
Indole-ynones as Privileged Substrates for Radical Dearomatizing Spirocyclization Cascades.
N. Inprung, H. E. Ho, J. A. Rossi-Ashton, R. G. Epton, A. C. Whitwood, J. M. Lynam, R. J. K. Taylor, M. J. James, W. P. Unsworth
Org. Lett. 2022, 24, 668–674
2021
63.
Unprecedented reactions for molecular editing.
William. P. Unsworth and Alyssa-Jennifer Avestro
Nature 2021, 593, 203–204
News and Views article
Unprecedented reactions for molecular editing.
William. P. Unsworth and Alyssa-Jennifer Avestro
Nature 2021, 593, 203–204
News and Views article
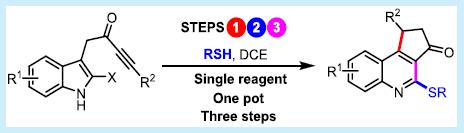
62.
A Thiol-Mediated Three-Step Ring Expansion Cascade for the Conversion of Indoles into Functionalized Quinolines.
Nantachai Inprung, Michael J. James,* Richard J. K. Taylor* and William. P. Unsworth*
Org. Lett. 2021, 23, 6, 2063–2068
A Thiol-Mediated Three-Step Ring Expansion Cascade for the Conversion of Indoles into Functionalized Quinolines.
Nantachai Inprung, Michael J. James,* Richard J. K. Taylor* and William. P. Unsworth*
Org. Lett. 2021, 23, 6, 2063–2068
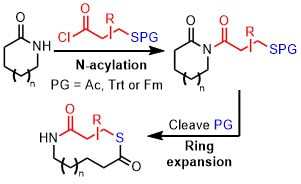
61.
Synthesis of macrocyclic and medium-sized ring thiolactones via the ring expansion of lactams
Kleopas Y. Palate, Ryan G. Epton, Adrian C. Whitwood, Jason M. Lynam and William P. Unsworth*
Org. Biolmol. Chem. 2021 https://doi.org/10.1039/D0OB02502J
Synthesis of macrocyclic and medium-sized ring thiolactones via the ring expansion of lactams
Kleopas Y. Palate, Ryan G. Epton, Adrian C. Whitwood, Jason M. Lynam and William P. Unsworth*
Org. Biolmol. Chem. 2021 https://doi.org/10.1039/D0OB02502J
2020
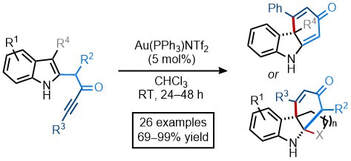
60.
Synthesis of polycyclic scaffolds via a gold-catalysed dearomative cyclisation cascade
Aimee K. Clarke,* James A. Rossi-Ashton, Richard J. K. Taylor and William P. Unsworth*
Tetrahedron 2020, https://doi.org/10.1016/j.tet.2020.131392
Synthesis of polycyclic scaffolds via a gold-catalysed dearomative cyclisation cascade
Aimee K. Clarke,* James A. Rossi-Ashton, Richard J. K. Taylor and William P. Unsworth*
Tetrahedron 2020, https://doi.org/10.1016/j.tet.2020.131392
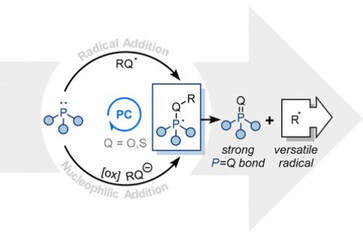
59.
Phosphoranyl Radical Fragmentation Reactions Driven by Photoredox Catalysis
James A. Rossi-Ashton*, Aimee K. Clarke, William P. Unsworth, and Richard J. K. Taylor
ACS Catal. 2020, 10, 7250-7261
Phosphoranyl Radical Fragmentation Reactions Driven by Photoredox Catalysis
James A. Rossi-Ashton*, Aimee K. Clarke, William P. Unsworth, and Richard J. K. Taylor
ACS Catal. 2020, 10, 7250-7261
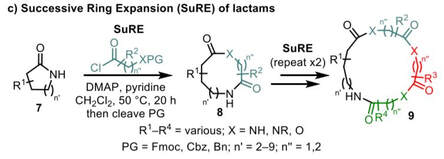
58.
Evaluating the viability of successive ring expansion reactions based on amino acid and hydroxyacid side chain insertion
Aggie Lawer, Ryan G. Epton, Thomas C. Stephens, Kleopas Y. Palate, Mahendar Lodi, Emilie Marotte, Katie J. Lamb, Jade K. Sangha, Jason M. Lynam,* William Paul Unsworth*
Chem. Eur. J., 2020, 26, 12674–12683
Evaluating the viability of successive ring expansion reactions based on amino acid and hydroxyacid side chain insertion
Aggie Lawer, Ryan G. Epton, Thomas C. Stephens, Kleopas Y. Palate, Mahendar Lodi, Emilie Marotte, Katie J. Lamb, Jade K. Sangha, Jason M. Lynam,* William Paul Unsworth*
Chem. Eur. J., 2020, 26, 12674–12683
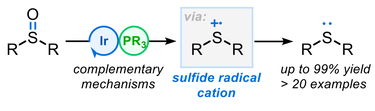
57.
Photocatalytic Deoxygenation of Sulfoxides using Visible Light: Mechanistic Investigations and Synthetic Applications
Aimee K Clarke, Alison Parkin, Richard J. K. Taylor*, William P. Unsworth* and James A. Rossi-Ashton*
ACS Catal. 2020, 10, 5814–5820
Photocatalytic Deoxygenation of Sulfoxides using Visible Light: Mechanistic Investigations and Synthetic Applications
Aimee K Clarke, Alison Parkin, Richard J. K. Taylor*, William P. Unsworth* and James A. Rossi-Ashton*
ACS Catal. 2020, 10, 5814–5820
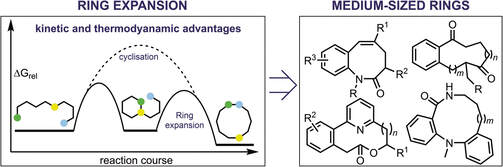
56.
A happy medium: the synthesis of medicinally important medium-sized rings via ring expansion
Aimee K Clarke and William P. Unsworth*
Chem. Sci. 2020, 11, 2876-2881
A happy medium: the synthesis of medicinally important medium-sized rings via ring expansion
Aimee K Clarke and William P. Unsworth*
Chem. Sci. 2020, 11, 2876-2881
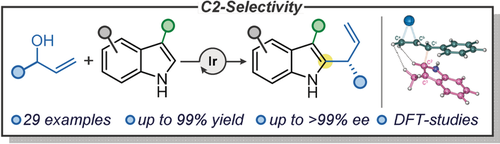
55.
Iridium Catalyzed Enantioselective Intermolecular Indole C2-Allylation
James A. Rossi-Ashton, Aimee K. Clarke, James R. Donald, Chao Zheng,*Richard J. K. Taylor,* William P. Unsworth,* Shu-Li You*
Angew. Chem. Int. Ed. 2020, 59, 7598–7604
Hot Paper
Iridium Catalyzed Enantioselective Intermolecular Indole C2-Allylation
James A. Rossi-Ashton, Aimee K. Clarke, James R. Donald, Chao Zheng,*Richard J. K. Taylor,* William P. Unsworth,* Shu-Li You*
Angew. Chem. Int. Ed. 2020, 59, 7598–7604
Hot Paper
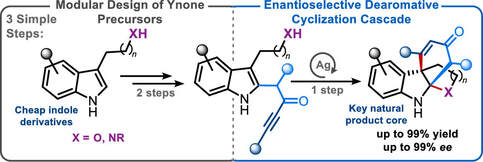
54.
Modular Synthesis of Polycyclic Alkaloid Scaffolds via an Enantioselective Dearomative Cascade
James A. Rossi-Ashton, Aimee K. Clarke, *Richard J. K. Taylor,* William P. Unsworth,*
Org. Lett. 2020, 22, 1175-1181
Modular Synthesis of Polycyclic Alkaloid Scaffolds via an Enantioselective Dearomative Cascade
James A. Rossi-Ashton, Aimee K. Clarke, *Richard J. K. Taylor,* William P. Unsworth,*
Org. Lett. 2020, 22, 1175-1181
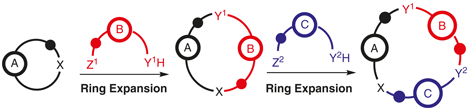
53.
Consecutive Ring-Expansion Reactions for the Iterative Assembly of Medium-Sized Rings and Macrocycles
Thomas C. Stephens and William P. Unsworth*
Synlett 2020, 31, 133-146
Consecutive Ring-Expansion Reactions for the Iterative Assembly of Medium-Sized Rings and Macrocycles
Thomas C. Stephens and William P. Unsworth*
Synlett 2020, 31, 133-146
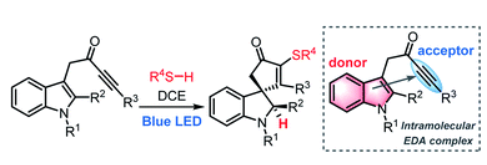
52.
Visible-light-induced intramolecular charge transfer in the radical spirocyclisation of indole-tethered ynones
Hon Eong Ho, Angela Pagano, James A. Rossi-Ashton, James R. Donald, Ryan G. Epton, Jonathan C. Churchill, Michael J. James, Peter O'Brien, Richard J. K. Taylor* and William P. Unsworth*
Chem. Sci., 2020,11, 1353-1360
Visible-light-induced intramolecular charge transfer in the radical spirocyclisation of indole-tethered ynones
Hon Eong Ho, Angela Pagano, James A. Rossi-Ashton, James R. Donald, Ryan G. Epton, Jonathan C. Churchill, Michael J. James, Peter O'Brien, Richard J. K. Taylor* and William P. Unsworth*
Chem. Sci., 2020,11, 1353-1360
2019
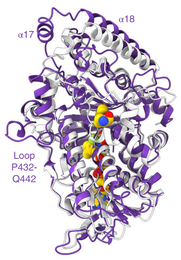
51. A Marine Viral Halogenase that Iodinates Diverse Substrates
D. S. Gkotsi, H. Ludewig, S. V. Sharma, W. P. Unsworth, R. J. K. Taylor, M. M. W. McLachlan, S. Shanahan, J. H. Naismith, R. J. M. Goss*
Nature Chem. 2019, 11, 1091–1097
D. S. Gkotsi, H. Ludewig, S. V. Sharma, W. P. Unsworth, R. J. K. Taylor, M. M. W. McLachlan, S. Shanahan, J. H. Naismith, R. J. M. Goss*
Nature Chem. 2019, 11, 1091–1097
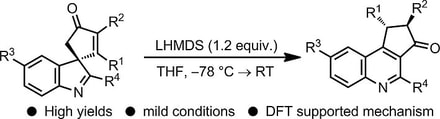
50.
Synthetic and Mechanistic Studies into the Rearrangement of Spirocyclic Indolenines into Quinolines
Ryan G. Epton, Aimee K. Clarke, Richard J. K. Taylor, William P. Unsworth* and Jason M. Lynam*
Eur. J. Org. Chem. 2019, 5563-5571
Synthetic and Mechanistic Studies into the Rearrangement of Spirocyclic Indolenines into Quinolines
Ryan G. Epton, Aimee K. Clarke, Richard J. K. Taylor, William P. Unsworth* and Jason M. Lynam*
Eur. J. Org. Chem. 2019, 5563-5571
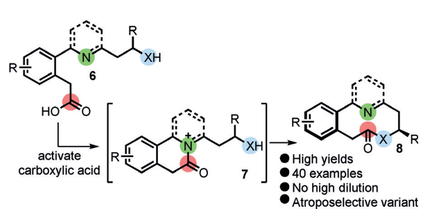
49.
Internal nucleophilic catalyst mediated cyclisation/ring expansion cascades for the synthesis of medium-sized lactones and lactams
Aggie Lawer James A. Rossi‐Ashton Thomas C. Stephens Bradley J. Challis Ryan G. Epton Jason M. Lynam and William P. Unsworth*
Angew. Chem. Int. Ed. 2019, 58, 13942-13947
Internal nucleophilic catalyst mediated cyclisation/ring expansion cascades for the synthesis of medium-sized lactones and lactams
Aggie Lawer James A. Rossi‐Ashton Thomas C. Stephens Bradley J. Challis Ryan G. Epton Jason M. Lynam and William P. Unsworth*
Angew. Chem. Int. Ed. 2019, 58, 13942-13947
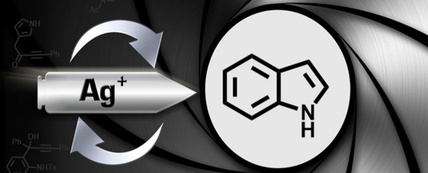
48.
Indole Synthesis Using Silver Catalysis
Aimee K. Clarke, Hon E. Ho, James A. Rossi‐Ashton, Richard J. K. Taylor and William P. Unsworth*
Chem. Asian J. 2019, 14, 1900-1911
Indole Synthesis Using Silver Catalysis
Aimee K. Clarke, Hon E. Ho, James A. Rossi‐Ashton, Richard J. K. Taylor and William P. Unsworth*
Chem. Asian J. 2019, 14, 1900-1911
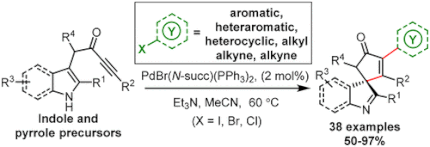
47.
Merging π-Acid and Pd Catalysis: Dearomatizing Spirocyclization/Cross-Coupling Cascade Reactions of Alkyne-Tethered Aromatics
Hon Eong Ho, Thomas C. Stephens, Thomas J. Payne, Peter O’Brien, Richard J. K. Taylor* and William P. Unsworth*
ACS Catal. 2019, 9, 1, 504-510
Merging π-Acid and Pd Catalysis: Dearomatizing Spirocyclization/Cross-Coupling Cascade Reactions of Alkyne-Tethered Aromatics
Hon Eong Ho, Thomas C. Stephens, Thomas J. Payne, Peter O’Brien, Richard J. K. Taylor* and William P. Unsworth*
ACS Catal. 2019, 9, 1, 504-510
2018
46.
Understanding the Role of Spiroindolenines in Pictet-Spengler Reactions
William P. Unsworth*
Chem. 2018, 4, 1767-1770
Understanding the Role of Spiroindolenines in Pictet-Spengler Reactions
William P. Unsworth*
Chem. 2018, 4, 1767-1770
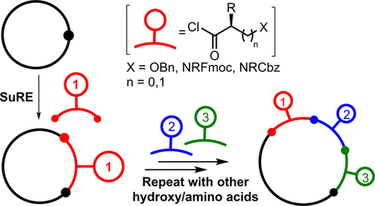
45.
Iterative Assembly of Macrocyclic Lactones using Successive Ring Expansion Reactions
Thomas C. Stephens, Aggie Lawer, Thomas French and William P. Unsworth*
Chem. Eur. J. 2018, 24, 13947-13953
Iterative Assembly of Macrocyclic Lactones using Successive Ring Expansion Reactions
Thomas C. Stephens, Aggie Lawer, Thomas French and William P. Unsworth*
Chem. Eur. J. 2018, 24, 13947-13953
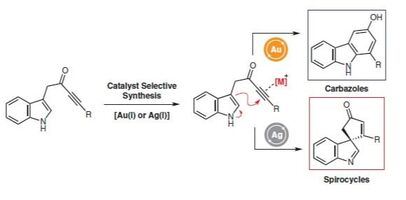
44.
Divergent reactivity of indole tethered ynones with silver(I) and gold(I) catalysis: a combined synthetic and computational study
J. T. R. Liddon, J. A. Rossi-Ashton, A. K. Clarke, J. M. Lynam, R. J. K. Taylor,* W. P. Unsworth* Synthesis 2018, 50, 4829–4836
Divergent reactivity of indole tethered ynones with silver(I) and gold(I) catalysis: a combined synthetic and computational study
J. T. R. Liddon, J. A. Rossi-Ashton, A. K. Clarke, J. M. Lynam, R. J. K. Taylor,* W. P. Unsworth* Synthesis 2018, 50, 4829–4836
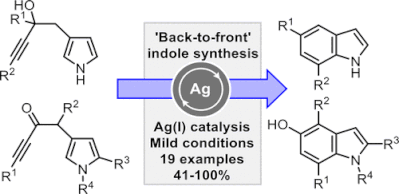
43.
"Back-to-Front" Indole Synthesis using Silver(I) Catalysis: Unexpected C-3 Pyrrole Activation Mode Supported by DFT
Aimee K. Clarke, Jason M. Lynam*, Richard J. K. Taylor* and William P. Unsworth*
ACS Catal. 2018, 8, 6844-6850
"Back-to-Front" Indole Synthesis using Silver(I) Catalysis: Unexpected C-3 Pyrrole Activation Mode Supported by DFT
Aimee K. Clarke, Jason M. Lynam*, Richard J. K. Taylor* and William P. Unsworth*
ACS Catal. 2018, 8, 6844-6850
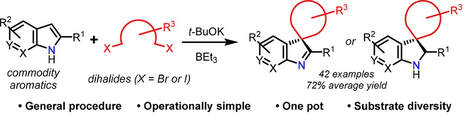
42.
Dearomatizing Spiroannulation Reagents: Direct Access to Spirocycles from Indoles and Dihalides
J. T. R. Liddon, J. A. Rossi-Ashton, R. J. K. Taylor, W. P. Unsworth
Org. Lett. 2018, 20, 3349−3353
Dearomatizing Spiroannulation Reagents: Direct Access to Spirocycles from Indoles and Dihalides
J. T. R. Liddon, J. A. Rossi-Ashton, R. J. K. Taylor, W. P. Unsworth
Org. Lett. 2018, 20, 3349−3353
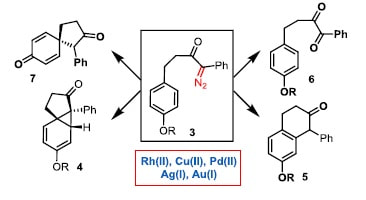
41.
Divergent reactivity of phenol- and anisole-tethered donor-acceptor α-diazoketones
A. K. Clarke, R. J. K. Taylor,* W. P. Unsworth,*
Tetrahedron, 2018, 74, 5374–5382
Divergent reactivity of phenol- and anisole-tethered donor-acceptor α-diazoketones
A. K. Clarke, R. J. K. Taylor,* W. P. Unsworth,*
Tetrahedron, 2018, 74, 5374–5382
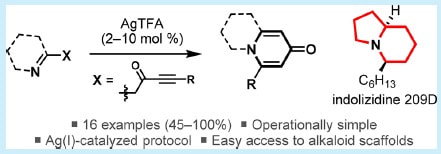
40.
Ag(I)-Catalyzed Synthesis of Azabicyclic Alkaloid Frameworks from Ketimine-Tethered Ynones: Total Synthesis of Indolizidine 209D
H. E. Ho, M. J. James, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Org. Lett. 2018, 20, 1439–1443
Ag(I)-Catalyzed Synthesis of Azabicyclic Alkaloid Frameworks from Ketimine-Tethered Ynones: Total Synthesis of Indolizidine 209D
H. E. Ho, M. J. James, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Org. Lett. 2018, 20, 1439–1443
2017
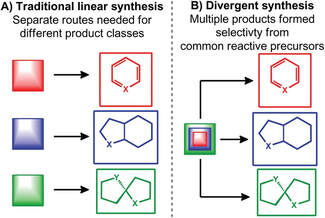
39.
Selective synthesis of three product classes from imine and carboxylic acid precursors via direct imine acylation
J. A. Rossi-Ashton, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem., 2017, 15, 7527
Selective synthesis of three product classes from imine and carboxylic acid precursors via direct imine acylation
J. A. Rossi-Ashton, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem., 2017, 15, 7527
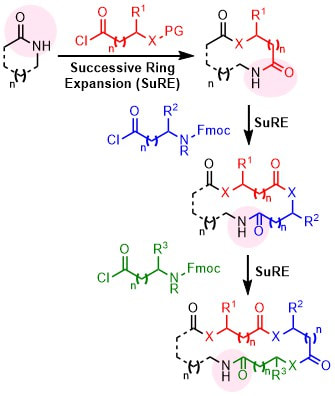
38.
Synthesis of Cyclic Peptide Mimetics by the Successive Ring Expansion of Lactams
T. C. Stephens, M. Lodi, A. Steer, Y. Lin, M. Gill, W. P. Unsworth*
Chem. Eur. J. 2017, 23, 13314–13318
Synthesis of Cyclic Peptide Mimetics by the Successive Ring Expansion of Lactams
T. C. Stephens, M. Lodi, A. Steer, Y. Lin, M. Gill, W. P. Unsworth*
Chem. Eur. J. 2017, 23, 13314–13318
37.
Ring-Expansion Reactions in the Synthesis of Macrocycles and Medium-Sized Rings
W. P. Unsworth,* J. R. Donald
Chem. Eur. J. 2017, 23, 8780
Ring-Expansion Reactions in the Synthesis of Macrocycles and Medium-Sized Rings
W. P. Unsworth,* J. R. Donald
Chem. Eur. J. 2017, 23, 8780
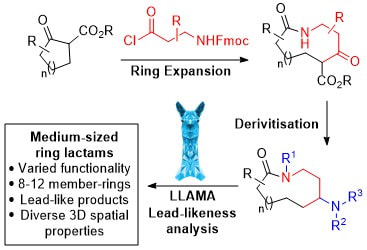
36.
Ring expansion approach to medium-sized lactams and analysis of their medicinal lead-like properties
L. G. Baud, M. A. Manning, H. L. Arkless, T. C. Stephens, W. P. Unsworth
Chem. Eur. J. 2017, 23, 2225.
Ring expansion approach to medium-sized lactams and analysis of their medicinal lead-like properties
L. G. Baud, M. A. Manning, H. L. Arkless, T. C. Stephens, W. P. Unsworth
Chem. Eur. J. 2017, 23, 2225.
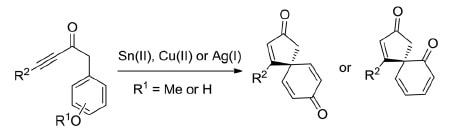
35.
Dearomatisation approaches to spirocyclic dienones via the electrophilic activation of alkynes
A. K. Clarke, J. T. R. Liddon, J. D. Cuthbertson, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem., 2017, 15, 233–245.
Dearomatisation approaches to spirocyclic dienones via the electrophilic activation of alkynes
A. K. Clarke, J. T. R. Liddon, J. D. Cuthbertson, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem., 2017, 15, 233–245.
2016
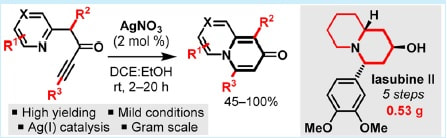
34.
Catalytic Dearomatization Approach to Quinolizidine Alkaloids: Five Step Total Synthesis of (±)-Lasubine II
M. J. James, N. D. Grant, Peter O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Org.Lett. 2016, 18, 6256−6259
Catalytic Dearomatization Approach to Quinolizidine Alkaloids: Five Step Total Synthesis of (±)-Lasubine II
M. J. James, N. D. Grant, Peter O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Org.Lett. 2016, 18, 6256−6259
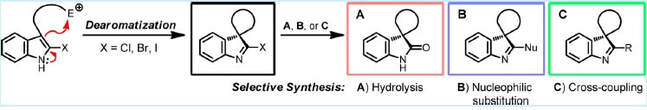
33.
Preparation and Reactions of Indoleninyl Halides: Scaffolds for the Synthesis of Spirocyclic Indole Derivatives
J. T. R. Liddon, A. K. Clarke, R. J. K. Taylor,* W. P. Unsworth*
Org. Lett. 2016, 18, 6328−6331
Preparation and Reactions of Indoleninyl Halides: Scaffolds for the Synthesis of Spirocyclic Indole Derivatives
J. T. R. Liddon, A. K. Clarke, R. J. K. Taylor,* W. P. Unsworth*
Org. Lett. 2016, 18, 6328−6331
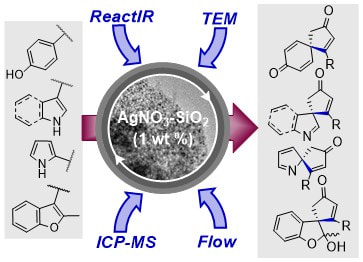
32.
Silica-supported silver nitrate as a highly active dearomatising spirocyclisation catalyst: synergistic alkyne activation by silver nanoparticles and silica
A. K. Clarke, M. J. James, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Angew. Chem. Int. Ed. 2016, 55, 13798–13802
Silica-supported silver nitrate as a highly active dearomatising spirocyclisation catalyst: synergistic alkyne activation by silver nanoparticles and silica
A. K. Clarke, M. J. James, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Angew. Chem. Int. Ed. 2016, 55, 13798–13802
31.
Phosphorylated cyclopropanes in the synthesis of α-alkylidene-γ-butyrolactones: total synthesis of (±)-savinin, (±)-gadain and (±)-peperomin E
M. G. Lloyd, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem. 2016, 14, 8971–8988
Phosphorylated cyclopropanes in the synthesis of α-alkylidene-γ-butyrolactones: total synthesis of (±)-savinin, (±)-gadain and (±)-peperomin E
M. G. Lloyd, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem. 2016, 14, 8971–8988
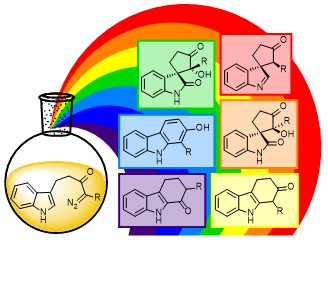
30.
Selective Synthesis of Six Products from a Single Indolyl α-Diazocarbonyl Precursor
M. J. James, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Angew. Chem. Int. Ed. 2016, 54, 9671–9675
Selective Synthesis of Six Products from a Single Indolyl α-Diazocarbonyl Precursor
M. J. James, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Angew. Chem. Int. Ed. 2016, 54, 9671–9675
29.
Direct Imine Acylation: A Versatile Method for the Synthesis of Nitrogen-Containing Heterocycles, Spirocycles and Natural Products
W. P. Unsworth,* R. J. K. Taylor*
Synlett 2016, 27, 2051–2064
Direct Imine Acylation: A Versatile Method for the Synthesis of Nitrogen-Containing Heterocycles, Spirocycles and Natural Products
W. P. Unsworth,* R. J. K. Taylor*
Synlett 2016, 27, 2051–2064
28.
Synthetic approaches to pallimamine and analogues using direct imine acylation
T. O. Ronson, C. Kitsiou, W. P. Unsworth, R. J. K. Taylor*
Tetrahedron 2016, 72, 6099–6106
Synthetic approaches to pallimamine and analogues using direct imine acylation
T. O. Ronson, C. Kitsiou, W. P. Unsworth, R. J. K. Taylor*
Tetrahedron 2016, 72, 6099–6106
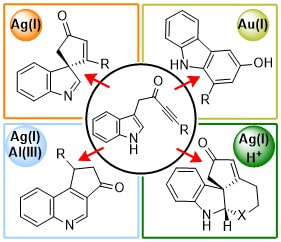
27.
Catalyst-Driven Scaffold Diversity: Selective Synthesis of Spirocycles, Carbazoles and Quinolines from Indolyl Ynones
J. T. R. Liddon, M. J. James, A. K. Clarke, P. O’Brien, R. J. K. Taylor, W. P. Unsworth
Chem. Eur. J. 2016, 22, 8777–8780.
Catalyst-Driven Scaffold Diversity: Selective Synthesis of Spirocycles, Carbazoles and Quinolines from Indolyl Ynones
J. T. R. Liddon, M. J. James, A. K. Clarke, P. O’Brien, R. J. K. Taylor, W. P. Unsworth
Chem. Eur. J. 2016, 22, 8777–8780.
26.
From Heteroaromatic Acids and Imines to Azaspirocycles: Stereoselective Synthesis and 3D Shape Analysis
S.J. Chambers, G. Coulthard, W. P. Unsworth, P. O’Brien, R. J. K. Taylor*
Chem. Eur. J. 2016, 22, 6496–6500
From Heteroaromatic Acids and Imines to Azaspirocycles: Stereoselective Synthesis and 3D Shape Analysis
S.J. Chambers, G. Coulthard, W. P. Unsworth, P. O’Brien, R. J. K. Taylor*
Chem. Eur. J. 2016, 22, 6496–6500
25.
A selective C–H insertion/olefination protocol for the synthesis of α-methylene-γ-butyrolactone natural products
M. G. Lloyd, M. D’Acunto, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem. 2016, 14, 1641–1645
A selective C–H insertion/olefination protocol for the synthesis of α-methylene-γ-butyrolactone natural products
M. G. Lloyd, M. D’Acunto, R. J. K. Taylor,* W. P. Unsworth*
Org. Biomol. Chem. 2016, 14, 1641–1645
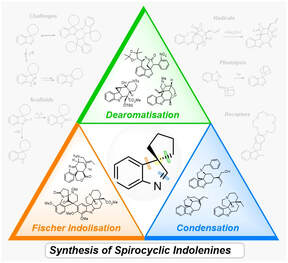
24.
Synthesis of Spirocyclic Indolenines
M. J. James, P. O’Brien, R. J. K. Taylor, W. P. Unsworth
Chem. Eur. J. 2016 22, 2856–2881
Synthesis of Spirocyclic Indolenines
M. J. James, P. O’Brien, R. J. K. Taylor, W. P. Unsworth
Chem. Eur. J. 2016 22, 2856–2881
23.
Synthetic Studies towards the Africanane Sesquiterpenes via the Cope Rearrangement of gem-Dimethyl-Substituted Divinyl Cyclopropanes
J. D. Osler, W. P. Unsworth,* R. J. K. Taylor*
Synlett, 2016, 27, 70–74
Synthetic Studies towards the Africanane Sesquiterpenes via the Cope Rearrangement of gem-Dimethyl-Substituted Divinyl Cyclopropanes
J. D. Osler, W. P. Unsworth,* R. J. K. Taylor*
Synlett, 2016, 27, 70–74
2015
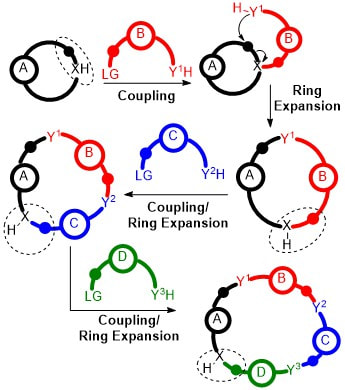
22.
The Synthesis of Structurally Diverse Macrocycles By Successive Ring Expansion
C. Kitsiou, J. J. Hindes, P. I’Anson, P. Jackson, T. C. Wilson, E. K. Daly, H. R. Felstead, P. Hearnshaw, W. P. Unsworth
Angew. Chem. Int. Ed. 2015, 54, 15794 –15798
The Synthesis of Structurally Diverse Macrocycles By Successive Ring Expansion
C. Kitsiou, J. J. Hindes, P. I’Anson, P. Jackson, T. C. Wilson, E. K. Daly, H. R. Felstead, P. Hearnshaw, W. P. Unsworth
Angew. Chem. Int. Ed. 2015, 54, 15794 –15798
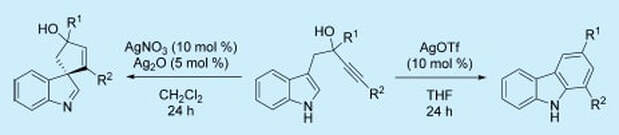
21.
Silver(I)-Catalyzed Dearomatization of Alkyne-Tethered Indoles: Divergent Synthesis of Spirocyclic Indolenines and Carbazoles
M. J. James, R. E. Clubley, K. Y. Palate, T. J. Procter, A. C. Wyton, P. O’Brien, R. J. K. Taylor, W. P. Unsworth
Org. Lett. 2015, 17, 4372
Silver(I)-Catalyzed Dearomatization of Alkyne-Tethered Indoles: Divergent Synthesis of Spirocyclic Indolenines and Carbazoles
M. J. James, R. E. Clubley, K. Y. Palate, T. J. Procter, A. C. Wyton, P. O’Brien, R. J. K. Taylor, W. P. Unsworth
Org. Lett. 2015, 17, 4372
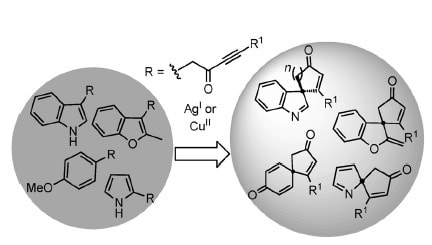
20.
Silver(I)- or Copper(II)-Mediated Dearomatization of Aromatic Ynones: Direct Access to Spirocyclic Scaffolds
M. J. James, J. Cuthbertson, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Angew. Chem. Int. Ed. 2015, 54, 7640–7643
Silver(I)- or Copper(II)-Mediated Dearomatization of Aromatic Ynones: Direct Access to Spirocyclic Scaffolds
M. J. James, J. Cuthbertson, P. O’Brien, R. J. K. Taylor,* W. P. Unsworth*
Angew. Chem. Int. Ed. 2015, 54, 7640–7643
19.
Propylphosphonic anhydride (T3P) mediated synthesis of β-lactams from imines and aryl-substituted acetic acids
G. Coulthard, W. P. Unsworth, R. J. K. Taylor*
Tetrahedron Lett, 2015, 56, 3113–3116
Propylphosphonic anhydride (T3P) mediated synthesis of β-lactams from imines and aryl-substituted acetic acids
G. Coulthard, W. P. Unsworth, R. J. K. Taylor*
Tetrahedron Lett, 2015, 56, 3113–3116
18.
The total synthesis of (+)-elaeokanidine A: natural product or isolation artefact?
J. D. Cuthbertson, W. P. Unsworth, C. L. Moody, R. J. K. Taylor*
Tetrahedron Lett, 2015, 56, 3123–3126
The total synthesis of (+)-elaeokanidine A: natural product or isolation artefact?
J. D. Cuthbertson, W. P. Unsworth, C. L. Moody, R. J. K. Taylor*
Tetrahedron Lett, 2015, 56, 3123–3126
17.
α-Alkylidene-γ-butyrolactone synthesis via one-pot C–H insertion/olefination: substrate scope and the total synthesis of (±)-cedarmycins A and B
M. G. Lloyd, M. D’Acunto, R. J. K. Taylor,* W. P. Unsworth*
Tetrahedron, 2015, 71, 7107–7123
α-Alkylidene-γ-butyrolactone synthesis via one-pot C–H insertion/olefination: substrate scope and the total synthesis of (±)-cedarmycins A and B
M. G. Lloyd, M. D’Acunto, R. J. K. Taylor,* W. P. Unsworth*
Tetrahedron, 2015, 71, 7107–7123
2014
16.
Rhodium(II)-catalysed tandem aziridination and ring-opening: stereoselective synthesis of functionalised tetrahydrofurans
W. P. Unsworth, T. Ronson, N. Clark, K. Stevens, A. L. Thompson, S. Lamont, J. Robertson*
Chem. Commun. 2014, 50, 11393–11396
Rhodium(II)-catalysed tandem aziridination and ring-opening: stereoselective synthesis of functionalised tetrahydrofurans
W. P. Unsworth, T. Ronson, N. Clark, K. Stevens, A. L. Thompson, S. Lamont, J. Robertson*
Chem. Commun. 2014, 50, 11393–11396
15.
Substrate scope and stereocontrol in the Rh(II)-catalysed oxyamination of allylic carbamates
W. P. Unsworth, S. G. Lamont, J. Robertson*
Tetrahedron 2014, 70, 7388–7394
Substrate scope and stereocontrol in the Rh(II)-catalysed oxyamination of allylic carbamates
W. P. Unsworth, S. G. Lamont, J. Robertson*
Tetrahedron 2014, 70, 7388–7394
14.
Substrate scope in the direct imine acylation of ortho-substituted benzoic acid derivatives: the total synthesis (±)-cavidine
C. Kitsiou, W. P. Unsworth, G. Coulthard, R. J. K. Taylor*
Tetrahedron 2014, 70, 7172–7180
Substrate scope in the direct imine acylation of ortho-substituted benzoic acid derivatives: the total synthesis (±)-cavidine
C. Kitsiou, W. P. Unsworth, G. Coulthard, R. J. K. Taylor*
Tetrahedron 2014, 70, 7172–7180
13.
A One-Pot C–H Insertion/Olefination Sequence for the Formation of α-Alkylidene-γ-butyrolactones
M. G. Lloyd, R. J. K. Taylor,* W. P. Unsworth*
Org. Lett. 2014,16, 2772
A One-Pot C–H Insertion/Olefination Sequence for the Formation of α-Alkylidene-γ-butyrolactones
M. G. Lloyd, R. J. K. Taylor,* W. P. Unsworth*
Org. Lett. 2014,16, 2772
12.
Direct Imine Acylation for Molecular Diversity in Heterocyclic Synthesis
W. P. Unsworth, G. Coulthard, C. Kitsiou, and R. J. K. Taylor*
J. Org. Chem., 2014, 79, 1368–1376
Direct Imine Acylation for Molecular Diversity in Heterocyclic Synthesis
W. P. Unsworth, G. Coulthard, C. Kitsiou, and R. J. K. Taylor*
J. Org. Chem., 2014, 79, 1368–1376
2013
11.
The Cope rearrangement of gem-dimethyl substituted divinylcyclopropanes
J. D. Osler, W. P. Unsworth, R. J. K. Taylor*
Org. Biomol. Chem. 2013, 11, 7587–7594
The Cope rearrangement of gem-dimethyl substituted divinylcyclopropanes
J. D. Osler, W. P. Unsworth, R. J. K. Taylor*
Org. Biomol. Chem. 2013, 11, 7587–7594
10.
‘Upenamide: trials and tribulations
W. P. Unsworth,* R. J. K. Taylor*
Org. Biomol. Chem. 2013, 11, 7250–7261
‘Upenamide: trials and tribulations
W. P. Unsworth,* R. J. K. Taylor*
Org. Biomol. Chem. 2013, 11, 7250–7261
9.
Total Synthesis of Spirobacillene A
W. P. Unsworth,* J. D. Cuthbertson, R. J. K. Taylor*
Org. Lett. 2013, 15, 3306–3309
Total Synthesis of Spirobacillene A
W. P. Unsworth,* J. D. Cuthbertson, R. J. K. Taylor*
Org. Lett. 2013, 15, 3306–3309
8.
An Expedient Protecting-Group-Free Total Synthesis of (±)-Dievodiamine
W. P. Unsworth,* C. Kitsiou, R. J. K. Taylor*
Org. Lett. 2013, 15, 3302–3305
An Expedient Protecting-Group-Free Total Synthesis of (±)-Dievodiamine
W. P. Unsworth,* C. Kitsiou, R. J. K. Taylor*
Org. Lett. 2013, 15, 3302–3305
7.
Direct Imine Acylation: Synthesis of the Proposed Structures of ‘Upenamide
W. P. Unsworth, K. A. Gallagher, M. Jean, J. P. Schmidt, L. J. Diorazio, R. J. K. Taylor*
Org. Lett. 2013, 15, 262–265
Direct Imine Acylation: Synthesis of the Proposed Structures of ‘Upenamide
W. P. Unsworth, K. A. Gallagher, M. Jean, J. P. Schmidt, L. J. Diorazio, R. J. K. Taylor*
Org. Lett. 2013, 15, 262–265
6.
Direct Imine Acylation: Rapid Access to Diverse Heterocyclic Scaffolds
W. P. Unsworth, C. Kitsiou, R. J. K. Taylor*
Org. Lett. 2013, 15, 258–261
Direct Imine Acylation: Rapid Access to Diverse Heterocyclic Scaffolds
W. P. Unsworth, C. Kitsiou, R. J. K. Taylor*
Org. Lett. 2013, 15, 258–261
2012 and earlier
5.
Alkyne-acetal cyclisation reactions mediated by formic acid; 3-acylated-2,5-dihydrofurans and related oxygen and nitrogen heterocycles
J. D. Cuthbertson, A. A. Godfrey, W. P. Unsworth, R. J. K. Taylor*
Heterocycles 2012, 84, 1013–1021
Alkyne-acetal cyclisation reactions mediated by formic acid; 3-acylated-2,5-dihydrofurans and related oxygen and nitrogen heterocycles
J. D. Cuthbertson, A. A. Godfrey, W. P. Unsworth, R. J. K. Taylor*
Heterocycles 2012, 84, 1013–1021
4.
Stereospecificity in the Au-catalysed cyclisation of monoallylic diols. Synthesis of (+)-isoaltholactone
W. P. Unsworth, K. Stevens, S. G. Lamont, J. Robertson*
Chem. Commun. 2011, 47, 7659–7661
Stereospecificity in the Au-catalysed cyclisation of monoallylic diols. Synthesis of (+)-isoaltholactone
W. P. Unsworth, K. Stevens, S. G. Lamont, J. Robertson*
Chem. Commun. 2011, 47, 7659–7661
3.
Aspects of stereocontrol in the L-Selectride reduction of 4-acyl-1,3-dioxolane derivatives
J. Robertson,* W. P. Unsworth, S. G. Lamont
Tetrahedron 2010, 66, 2363
Aspects of stereocontrol in the L-Selectride reduction of 4-acyl-1,3-dioxolane derivatives
J. Robertson,* W. P. Unsworth, S. G. Lamont
Tetrahedron 2010, 66, 2363
2.
Radical 1,4-aryl transfer in arylcarboxamides leading to phthalimides, biaryls and enantiomerically enriched β-arylethylamines
J. Robertson,* M. J. Palframan, S. A. Shea, K. Tchabanenko, W. P. Unsworth, C. Winters
Tetrahedron 2008, 64, 11896–11907
Radical 1,4-aryl transfer in arylcarboxamides leading to phthalimides, biaryls and enantiomerically enriched β-arylethylamines
J. Robertson,* M. J. Palframan, S. A. Shea, K. Tchabanenko, W. P. Unsworth, C. Winters
Tetrahedron 2008, 64, 11896–11907
1.
Further studies on silatropic carbonyl ene cyclisations: β-crotyl(diphenyl)silyloxy aldehyde substrates; synthesis of 2-deoxy-2-C-phenylhexoses
J. Robertson,* S. P. Green, M. J. Hall, A. J. Tyrrell, W. P. Unsworth
Org. Biomol. Chem. 2008, 6, 2628–2635
Further studies on silatropic carbonyl ene cyclisations: β-crotyl(diphenyl)silyloxy aldehyde substrates; synthesis of 2-deoxy-2-C-phenylhexoses
J. Robertson,* S. P. Green, M. J. Hall, A. J. Tyrrell, W. P. Unsworth
Org. Biomol. Chem. 2008, 6, 2628–2635
